Finished pharmaceutical products including medicines, vaccines, biologics, injectables, specialised therapies (gene therapy, etc.), or even active pharmaceutical ingredients (APIs) lose their effectiveness and may be rendered unsafe in extreme temperatures. Hence, cold storage for pharmaceuticals is critical when it comes to maintaining the quality and integrity of these indispensable products.
Undoubtedly, not adhering to cold storage guidelines can risk the reputation and business of pharmaceutical companies.
Cold Storage for Pharmaceuticals: What You Need to Know
It’s been just over the last few years that the pharmaceutical industry has become a major consumer of the cold chain. There is a need for a constant and uniform temperature-controlled environment for drugs and related products, from the place of manufacture to transportation, handling, storage, and delivery.
This can be attributed to the new wave of innovation and development in specialised therapies. Many modern biologic therapies, monoclonal antibodies and vaccines, gene and cellular therapies, some forms of cancer treatment and certain types of insulin need a reliable infrastructure for maintaining the right temperature range for a prolonged period until they reach the patient. It is primarily important for the efficacy, safety, and shelf-life of these sensitive drugs.
Additionally, the drug manufacturing process has evolved with the inclusion of high-value ingredients. Such ingredients have a shorter shelf-life and stringent temperature requirements during storage.
It won’t be, thus, wrong to say that the need for temperature control and monitoring has increased considerably throughout the pharmaceutical supply chain.
In the pharmaceuticals industry, cold chain infrastructure and services include temperature-controlled storage at ambient and refrigerated conditions as well as frozen, ultra-frozen, vapour-phase liquid nitrogen and liquid-phase liquid nitrogen conditions.
Are There Specific Regulations on Medicine Storage?
Indeed! The United States Pharmacopeia (USP) has provided definitions on both medicine storage and shipment.
- Freezer: Temperature is controlled between −25° and −10°C (−13° and 14°F). In case storage condition below −20°C (−4°F) is required, the temperature of the storage place should be controlled to ±10°C.
- Refrigerator: Temperature is controlled between 2° and 8°C (36° and 46°F).
- Cold: Any temperature not over 8°C (46°F).
- Cool: Any temperature between 8° and 15°C (46° and 59°F).
- Room temperature (also known as ambient temperature): The temperature in a working environment.
- Controlled room temperature: The temperature maintained thermostatically that includes the usual and customary working environment of 20°–25°C (68°–77°F). Deviations or excursions between 15° and 30°C are permitted (temperatures that are experienced in pharmacies, hospitals, warehouses and during shipping).
Benefits of Cold Storage in the Pharmaceuticals Industry
Here are some of the proven benefits of using cold storage in pharmaceuticals.
-
Versatile storage
Cold storage can store and preserve a variety of temperature-sensitive pharmaceutical products, be it vaccines, drugs, or biologics. Cold rooms can be customised for capacity, dimensions, and temperature regulations based on business needs. Since these facilities are durable and have a longer service life, they provide more stable storage conditions with a reduced risk of exposure. This can help reduce the wastage of drugs and related products and also reduce the need for repurchasing.
-
Better use of space
Since cold rooms can be customised for various sizes, they can also meet demands when it comes to the utilisation of limited space. Depending on the space availability, refrigeration systems can be placed inside or outside for designing smart cold storage solutions. Moreover, effective racking solutions can also be a space saver in your cold rooms!
-
Energy savings
Smartly designed col storage facilities can also help you save on energy bills. Air-tight insulation for walls, doors, and ceilings, an energy-efficient refrigeration system, and temperature-controlling capability can help you avoid sudden temperature drops or changes. No doubt, smooth operations and regular maintenance can lead to long-term energy savings.
How Can Rinac Support You in the Healthcare Industry
Rinac, a frontrunner in cold chain and modular construction solutions has been partnering with pharamceutical companies, distributors, and hospitals to offer expert solutions.
In the healthcare industry, Rinac offers the following:
- Walk-in temperature and humidity-controlled stability chambers for accelerated testing of pharmaceutical products with data acquisition in compliance with CFR-21
- Low-temperature clean freezer rooms and cold rooms with flame-proof fittings (inclusive of data acquisition and retrieval)
- Select racking, mobile racking, and mezzanine facility in cold storage rooms
- Docking stations
- Air-cooled or water-cooled freon-based refrigeration system
- Ammonia-based refrigeration system
- Refrigerated transport vehicles
Apart from that, we also offer
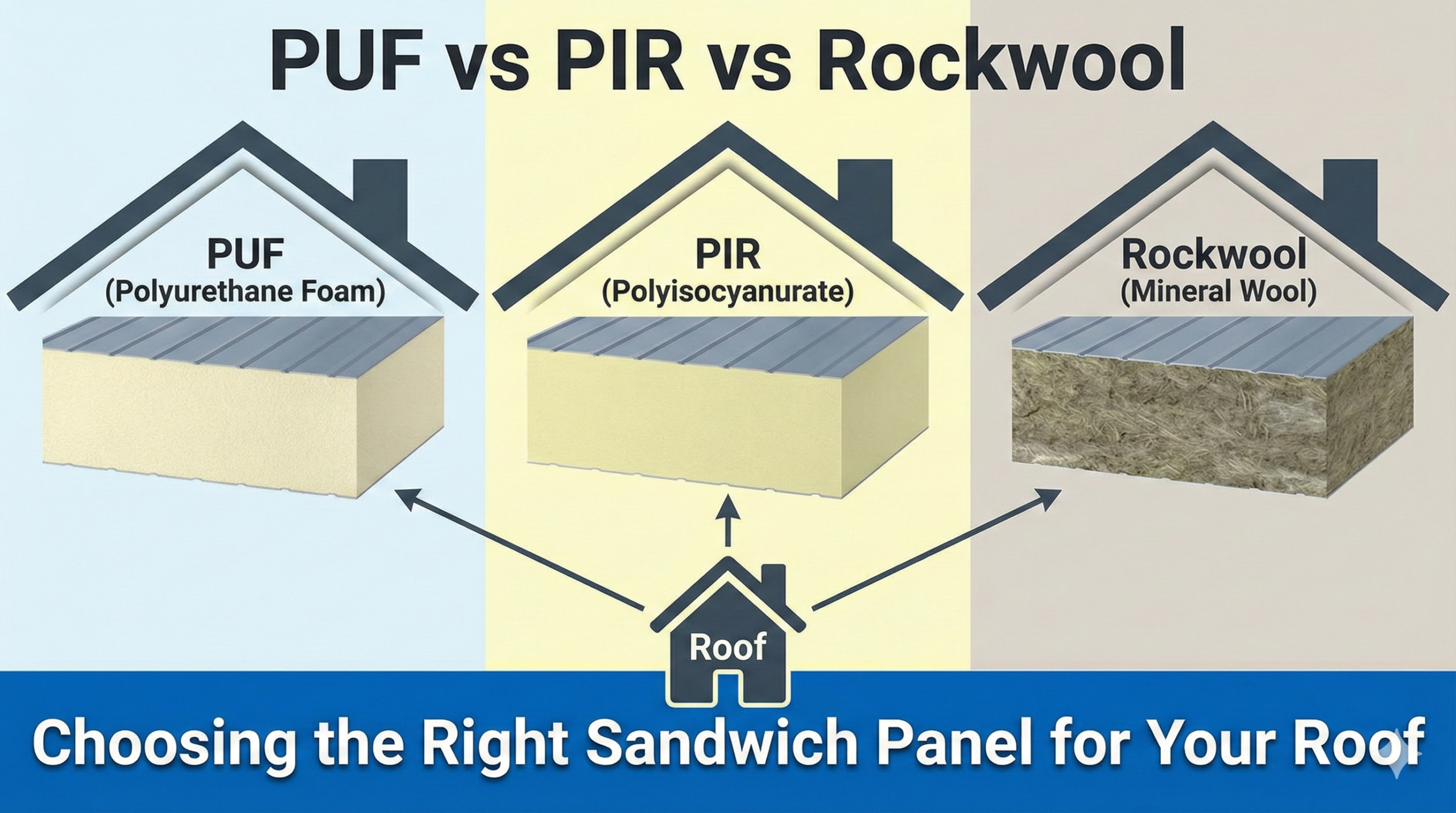
- Clean-zone and class 100000 constructions
- Clean-zone construction with RPFU/PIR sandwich panels, walls, ceilings, and doors which meet HACCP requirements
To know more about our solutions and how we can help your business, get in touch with us today!